
SOLVED: Find the final volume, in mL, of each of the following examples: A 1.5 M HCl solution prepared from 20.0 mL of a 6.0 M HCl solution A 2.0 % (m/v)

Two solutions labelled as 3 M HCl and 1 M HCl are mixed in the ratio of x:y by volume and the molarity of mixture solution becomes 1.5 M. What is the

480 mL of 1.5 M HCl was mixed with 520 Ml of 1.2 M HCl. What is the molarity of resulting solution ?

1 M HCl and 2 M HCl are mixed in volume ratio of 4: 1. The resulting HCl solution is diluted to double its original volume. What is the final molarity of
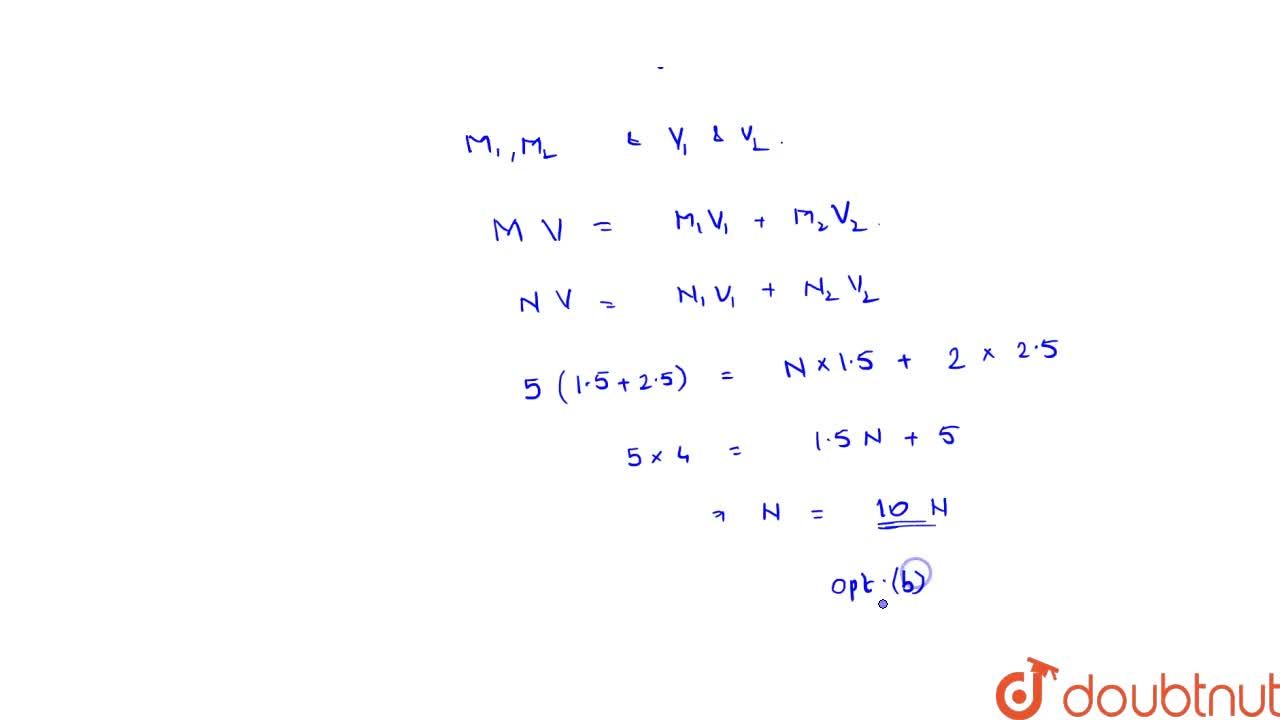
1.5 litre of a solution of normality N and 2.5 litre of 2 M HCl are mixed together. The resutant solution had a normality 5. The value of N is

Molar Mass & Molarity. Molar Mass Mass in grams of one mole of an element or compound. Numerically equal to the atomic weight of the element or the sum. - ppt download

SOLVED: How many mL of 1.5 M HCl solution is required to completely neutralize 20.00 mL of 10.0% calcium hydroxide solution (you can assume density is 1.00 g/mL), please show your work

A drop ( 0.05 mL) of 12M HCl is spread over a thin sheet of aluminium foil (thickness 0.10 mm and density of Al = 2.70 g/mL). Assuming whole of the HCl

SOLVED: Compute for the molarity of the solution when you mix 0.05 L of 2 M hydrochloric acid solution and 0.015 L of 1.5 M hydrochloric acid solution.
The volume of 1.5 M HCl required to completely react with 2.4 g Mg is - Sarthaks eConnect | Largest Online Education Community













![BT021-1] 1.5M Tris-HCl, pH 8.8 | Biosolution BT021-1] 1.5M Tris-HCl, pH 8.8 | Biosolution](http://biosolution.cafe24.com/wp-content/uploads/2015/05/BT021-1-1-5M-Tris-8-8.jpg)