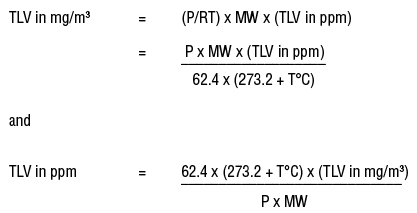
10.30 mg of O2 is dissolved into a liter of sea water of density 1.03 g/mL . The concentration of O2 in ppm is .

SOLVED: Convert 50 ppm to gL 19. Convert 50 ppm to mgL 20. Convert 5 ppb to uVI 21 Convert 3 ppm to ulml 22. Convert 350 ppm to mLL 23.300 ppb Cadmium (solid)

On litre of sea water weighs 1050 grams and contains 6 xx 10^(-3)g of dissolved oxygen gas. Calculate the concentration of the dissolved oxygen in ppm.

White Board Review Practicing with Concentration Expressions Molarity Percent ppm © Mr. D. Scott; CHS. - ppt download











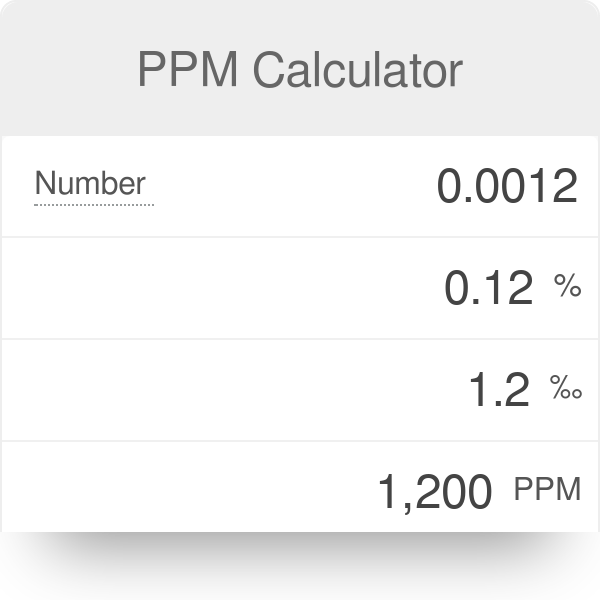
:max_bytes(150000):strip_icc()/how-to-calculate-normality-609580final2-0d5efa5a961f4fa0a7efc780921faee1.png)