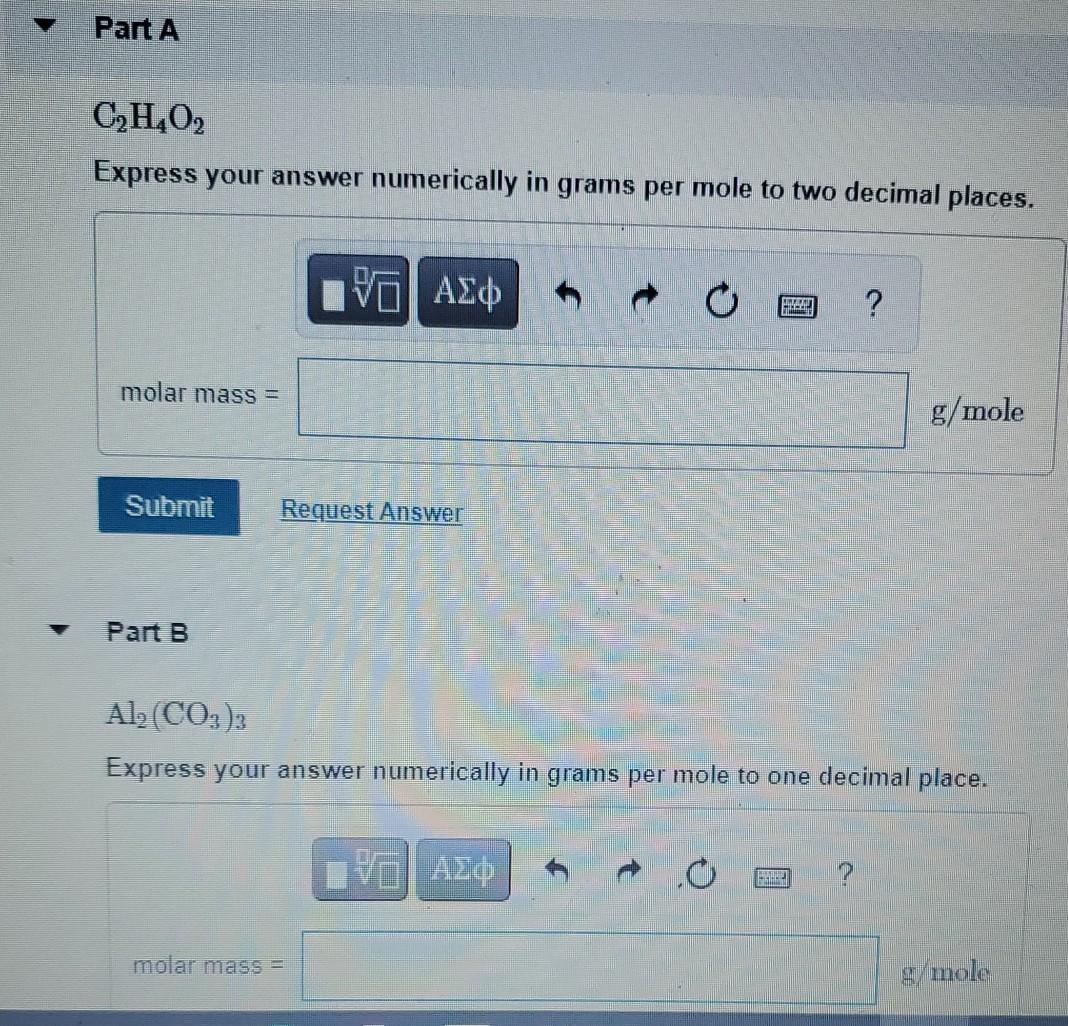
A compound has a molar mass of 90. grams per mole and the empirical formula CH2O. What is the - Brainly.com

SOLVED: What is the molecular formula for a compound with an empirical formula of C2OH4 and a molar mass of 88 grams per mole

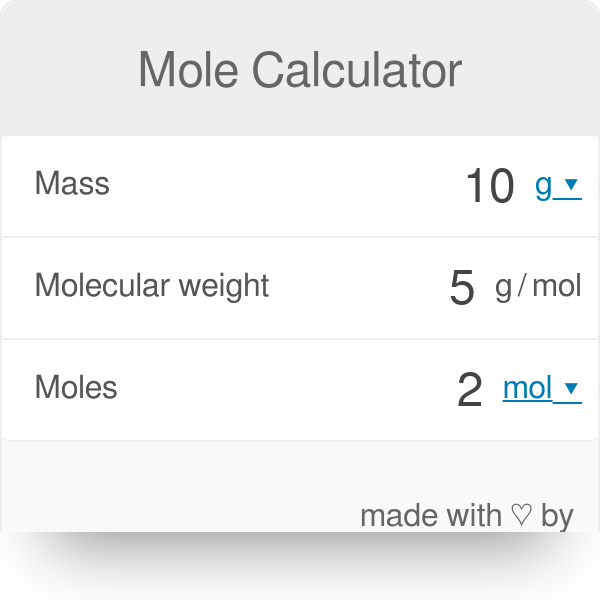
The molar mass of water is 18.0 grams per mole. What is the mass of 9.00 moles of water? - Brainly.com

Lecture 5. THE MOLE Avogadro's number The mole is used when we're talking about numbers of atoms and molecules (tiny particles).moleatomsmolecules The. - ppt download