For a complex reaction A k products Ea1 = 180 kJ/mol ; Ea2 = 80 kJ/mol ; Ea3 = 50 kJ/mol Overall rate constant k is related to individual rate constant by

Writing Rate Laws of Reaction Mechanisms Using The Rate Determining Step - Chemical Kinetics - YouTube

How to Combine a Series of Elementary Reactions into an Overall Balanced Equation | Chemistry | Study.com

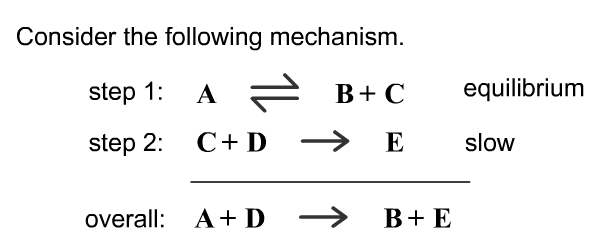
SOLVED: Consider the mechanism Step 1: A = B+C Step 2: C+D-E Overall: A +D - B+E equilibrium slow Determine the rate law for the overall reaction, where the overall rate constant

A chemical reaction proceeds as follows: A + C rarr D + F (Slow) F + B rarr G (Fast) G + C rarr E (Fast) Suggest rate law,units for rate constant,

Determine the rate law for the overall reaction (where the overall rate constant is represented as k) - Home Work Help - Learn CBSE Forum

Determine the rate law for the overall reaction (where the overall rate constant is represented as k)? | Socratic

The overall order of the reaction is the sum of the exponents of all the reactants in the rate expression.